>
Lilly
SKU: 56676
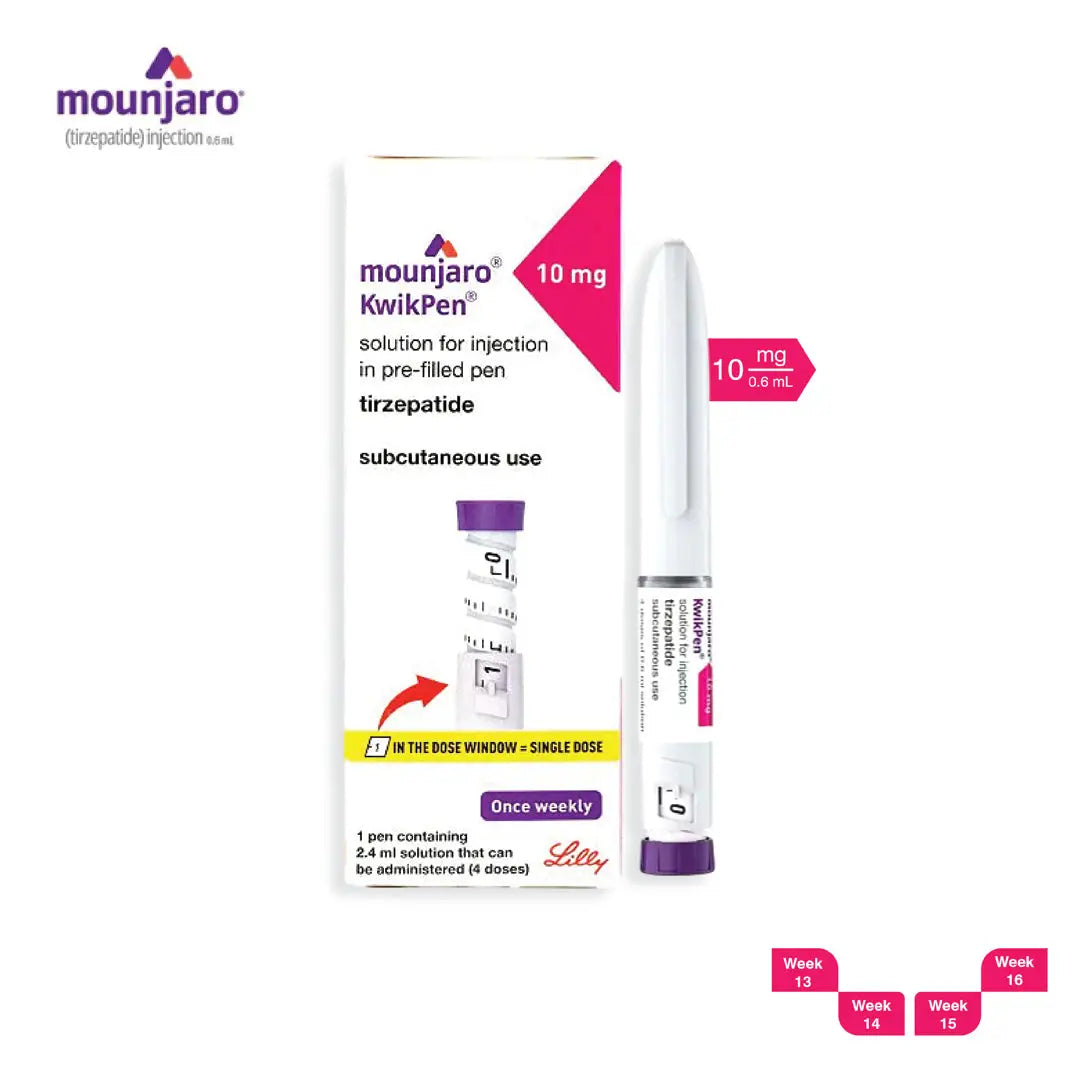
Mounjaro is a once-weekly, subcutaneous injection that contains tirzepatide, a dual GIP/GLP-1 receptor agonist. The pre-filled, single-use pen is designed for at-home administration with a simple, discreet injection experience when used exactly as prescribed.
Indications:
- Support weight reduction when combined with diet and exercise.
- Improve blood-sugar control in adults with type 2 diabetes, when used along with a balanced diet and regular physical activity.
Main Ingredient:
- Tirzepatide
Note:
- Does not contain pins for injection.
Indications:
- Support weight reduction when combined with diet and exercise.
- Improve blood-sugar control in adults with type 2 diabetes, when used along with a balanced diet and regular physical activity.
Main Ingredient:
- Tirzepatide
Note:
- Does not contain pins for injection.
Tirzepatide, Sodium Chloride, Sodium Phosphate Dibasic Heptahydrate, Water for injection, Hydrochloric acid solution and/or sodium hydroxide.
Recently Viewed